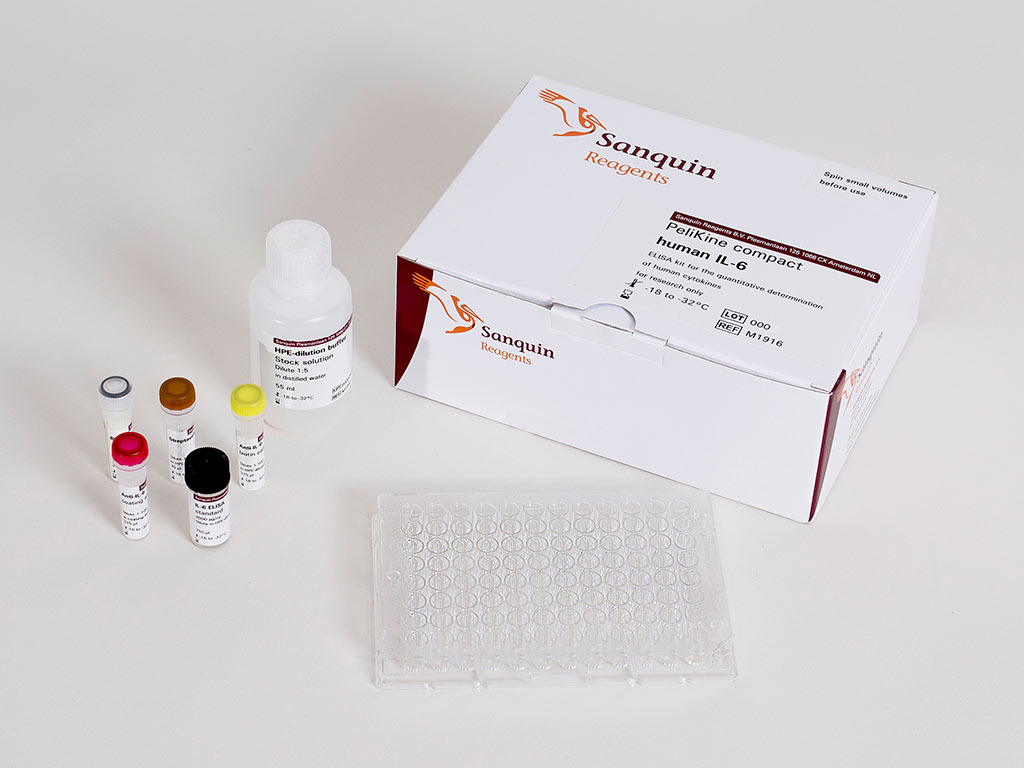
The PeliKine compact human IL-6 ELISA Kit is the optimal tool for sensitive detection of the pro-inflammatory cytokine Interleukin 6 (IL-6) released by human monocytes during exposure to endotoxins or pyrogens in the Monocyte Activation Test (MAT). IL-6 is a mediator of inflammatory responses and is involved in the development of fever.
The PeliKine compact human IL-6 kit is a "sandwich-type" of enzyme immunoassay in which a monoclonal antibody is pre-coated to polystyrene microtiter wells. The target cytokine, present in a measured volume of sample or standard, is bound by the antibody on the microtiter plate. Non-bound material is then removed by washing. Subsequently, a biotinylated antibody is added. This antibody binds to the cytokine-antibody complex present in the microtiter well. Excess biotinylated antibody is removed by washing, followed by addition of horseradish peroxidase (HRP) conjugated streptavidine, which binds onto the biotinylated side of the cytokine sandwich. After removal of non-bound HRP conjugate by washing, substrate solution is added to the wells. A colored product is formed in proportion to the amount of cytokine present in the sample or standard. After the reaction has been terminated by the addition of a stop solution, absorbance is measured in an absorbance reader at 450 nm.
The PeliKine compact human IL-6 ELISA kit has been validated for use with the PyroCell™ MAT Kit to conduct the MAT assay. All components together represent the PyroCell™ MAT System, IL-6 supporting the complete testing of up to 9 product sample preparations according to the compendial requirements of the European Pharmacopeia (EP 2.6.30). The sensitivity of the assay system is ? 0.02 EU/mL.
The Monocyte Activation Test (MAT) is an in vitro assay for pyrogen testing in the biopharmaceutical or medical device industry. The MAT test principle is based on the detection of inflammatory cytokines released by human monocytes in response to exogenous endotoxins (LPS) and non-endotoxin pyrogens (NEP).
An MAT assay is performed in two steps: during an overnight cell culture step, a monocytic cell preparation, e.g. peripheral blood mononuclear cells (PBMC) is stimulated for release of inflammatory cytokines with a product preparation that may contain a pyrogenic contaminant. On the next day, the cell culture supernatant is analyzed for the presence of released cytokines using an ELISA assay. The color reaction is measured in an absorbance reader, e.g. the ELx808™ Reader, at a wavelength of 450 nm and a reference wavelength of 540-590 nm. The pyrogenic content of the sample is then analyzed against the reference standard endotoxin (RSE) curve and is expressed in Endotoxin Equivalent Units (EEU/mL).
Endotoxin and Pyrogen Testing Products
The next chapter in endotoxin testing... Let's write it together.