For decades, you have relied on us for our trusted endotoxin testing reagents, consumables, devices, services and software to meet the ever-changing needs of regulated manufacturing environments.
Endotoxin automation can improve your lab efficiency and enhance compliance. Implementing the PyroTec® PRO Automated Robotic Solution in your laboratory can reduce the potential for human error substantially, and enhance the accuracy, reliability and traceability of results.
The capability for both rFC automation and Kinetic LAL automation (chromogenic and turbidimetric) on the PyroTec® PRO System is a significant industry development for BET. The versatility provided by Lonza’s proprietary systems means you can have the freedom to utilize the right test for all of your sample testing needs.
Integrated with WinKQCL® Analysis and Detection Software, the Lonza PyroTec® PRO Robotic System can streamline the methods, devices, training, data management, auditing, trending and servicing of your complete endotoxin testing program for the long run.
Lonza WinKQCL® Software version 6.3 PyroTec® PRO System Software module is integrated with a LoadingID on deck barcode scanning module which can greatly strengthen data integrity and efficiency in endotoxin assay automation.
Request a demoOverall Potential Benefits of Endotoxin Automation with the PyroTec® PRO System
Improve quality
- Improve control of QC processes
- Reduce manual pipetting errors
- Improve reproducibility
Prepare for the future
- Scale with business growth and change
- Realize more efficient testing
- Employ sustainable testing method options
Save time
- Reduce training burden
- Reduce revalidation requirementIncrease productivity
- Maintain existing work flows
Contact us to learn how BET automation can improve your QC testing program, together.

Streamlining Your QC Testing: Automated Endotoxin Testing and Process Optimization
View our webinar in our QC Insider® Toolbox to learn how automating the liquid handling aspect of the endotoxin test helps you save time and reduce manual pipetting errors and retests.
With the release of WinKQCL® Software version 6.3 the following PyroTec® PRO System enhancements are available:
Pyrotec® PRO System Update 2022 |
|
| Description | Type |
|
Support added for the LoadingID module for the PyroTec® PRO System |
Enhancement |
|
Introduction of a re-optimized new deck layout for the PyroTec® PRO System |
Enhancement |
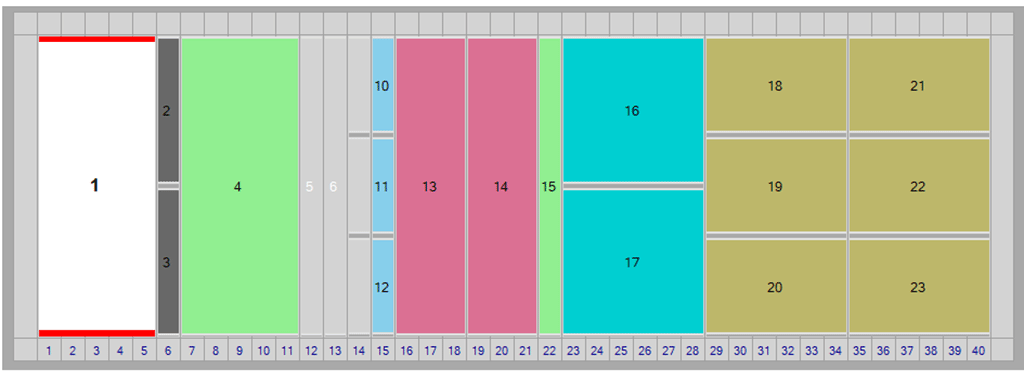
What is LoadingID?
LoadingID is an on deck 1D barcode scanner mounted to the left side of the PyroTec® PRO System deck. The LoadingID hardware and updated PyroTec® PRO System Software module together determine sample location in the LoadingID tube runners by reading 1D barcode labels on sample tubes and cross-referencing with the selected WinKQCL® Software template(s).
LoadingID can enhance data integrity by:
1. Ensuring the correct samples are picked, processed and plated correctly
2. Detecting duplicate samples
3. Alerting users to missing samples and unreadable barcode labels ensuring all samples in the selected WinKQCL® Software template are included in the run
Analysts can now place the samples anywhere in the LoadingID sample runners and the scanned barcode tells the software where each sample is located on which rack.
The Loading unit can be integrated into a new system at time of purchase or by retrofitting an existing PyroTec® PRO System. For retrofitting please note that the deck layout must be re-optimised and system re-qualification will be required.
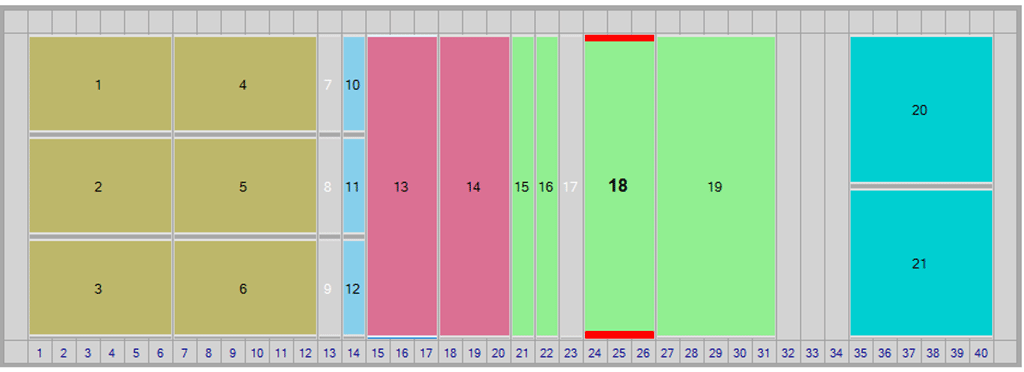
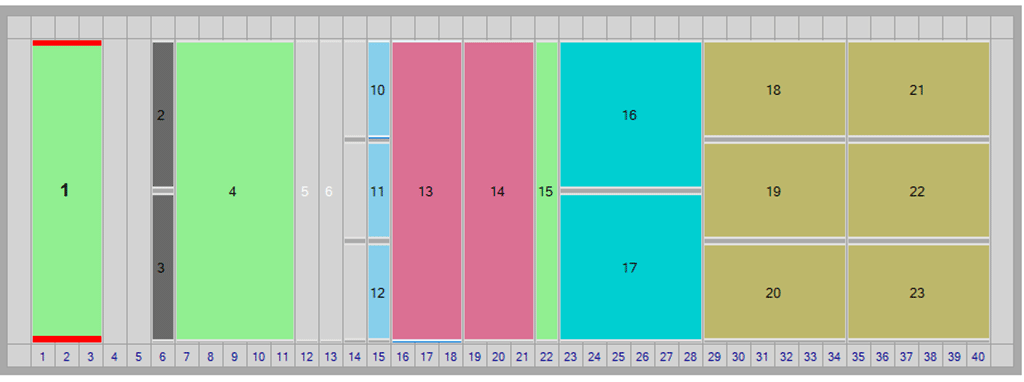
Optimized Deck Layout
In response to customer feedback we have recently optimised the layout to achieve greater efficiency in plate preparation for BET automation. The optimised deck layout includes movement of the samples (white for LoadingID or green for standard deck) and dilution tubes (green) to the far left of the deck and the tips (brown) and plate decks (blue) to the far right away from the CSE and reagents which remain in the centre of the deck.
Upgrade to WinKQCL® Software version 6.3 and the updated PyroTec® PRO System Software module is available for download at no charge for existing WinKQCL® Software v6 and PyroTec® PRO System customers. Please contact your local sales account manager to arrange quotation for any hardware upgrade and system re-qualification. For more information about WinKQCL® Software version 6.3 and the 2022 PyroTec® PRO System updates for endotoxin assay automation please contact our trusted Scientific Support Team.
When planning the implementation of endotoxin automation with a PyroTec® PRO System in your facility, allocation and scheduling of responsible persons as early as possible in the process is critical to success. In particular, assigning a project manager to coordinate efforts, IT resource for software installation and configuration and QA resource for documentation review and system go live sign off impact right first time project completion.
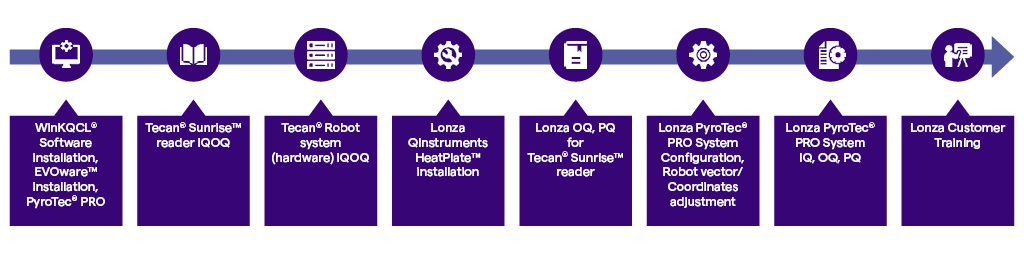
Our trained and certified engineers perform the system IQOQPQ following a specific sequence governed by controlled documentation for the building, testing and qualification of the mechanical, electronic and software components:
Once on site engineers uncrate and assemble the robot which is followed by IQ and OQ testing of the performance of the readers and robotic system components. The PyroTec® PRO System configuration specific for the endotoxin automation application (kinetic LAL automation and/or rFC automation) commences with IQOQPQ of the on deck devices and labware. The robotic arm movements are fine-tuned to enable optimal alignment and performance. A standardized PQ assay protocol is conducted to test the full functionality of the system for each assay method for BET automation.
It is recommended that you prepare your own PQ protocol for endotoxin automation based on knowledge of the types of samples to be tested and the results obtained by the current manual BET method.
The PyroTec® PRO System is both simple to operate and to maintain. Users carry out a few simple procedures, through the WinKQCL® Software, at specified intervals to ensure efficient daily operation. The system can be protected by purchasing a Lonza maintenance contract that provides for both routine annual maintenance and emergency call out in the event of breakdown. Troubleshooting and support is accessed through our trusted Scientific Support Team.
Lonza can assist evaluation of a customer’s current workflow using return on investment (ROI) modelling to support business case proposal for endotoxin automation with the PyroTec® PRO System. Details such as sample throughput, repeat testing rates, shift rotations, methods in use, sample types and manipulations required and future growth will all influence a successful ROI determination. Aspects such as enhanced data integrity, reduction in repetitive strain injury risk, and simplified end user training needs should also be considered as part of any business case for endotoxin automation.
The example in the table highlights the estimated time-savings that can be achieved by endotoxin automation with the Lonza PyroTec® PRO Automated Robotic Solution.
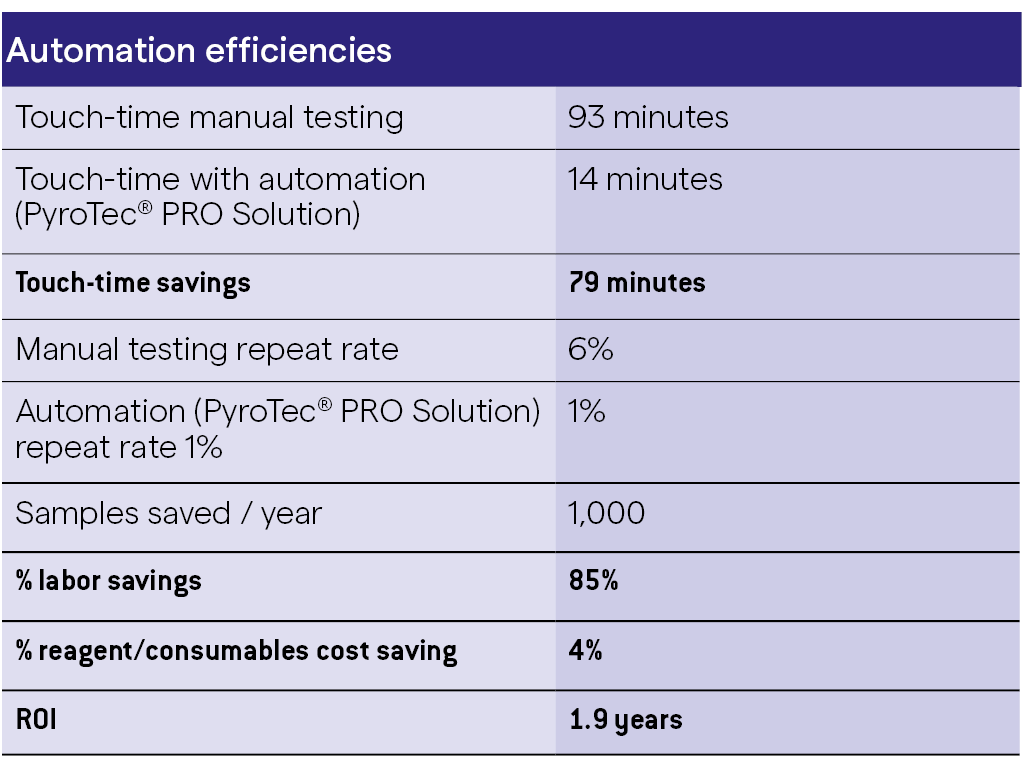
This table illustrates the estimated time-savings, reduction in repeat testing rate and consumables cost savings for a laboratory testing 20,000 water samples per annum using a manual kinetic chromogenic plate-based method including interference controls (positive product control, PPC) with each sample. The Return on Investment for this example is 1.9 years, well below the typically targeted 3 year ROI.
Learn more in our Whitepaper “Automation and Digitalization, The Key to Boosting Your Lab’s Efficiency, Productivity and Quality”.
We realize the budget process requires ample justification for your investment in endotoxin automation; our sales team are available to help support and justify your time and monetary investment.
Let’s see how automation will improve your endotoxin testing program, together.
Please contact our Scientific Support Team to start exploring a business case for endotoxin automation in your organization.
Ethicon’s Road to QC Automation
New White Paper Published in our QC Inside® Toolbox
Learn how Lonza worked with medical device company Ethicon to successfully build a business case and implement the Lonza PyroTec® PRO Automated Robotic Solution for efficient, QC operations that help prepare for the future.
Login or subscribeAutomating the BET with Dynamic Scripting
White Paper Available in our QC Insider® Toolbox
Learn how the PyroTec PRO® Robotic System can easily accommodate process changes and fully automate the endotoxin assay, reduce plating and pipetting errors, and allow analysts more time to perform higher value tasks.
Login or subscribeLonza’s PyroTec® PRO System
See It in Action
Contact us to schedule an in person or virtual demonstration of the Lonza PyroTec® PRO System. Test your samples and discuss with our experts how endotoxin automation will work in your lab. We have demonstration systems located at sites in the US, Europe and Asia Pacific.
Request a demo